You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
HEALTH “Catastrophic Contagion” - Gates newest tabletop exercise (OP Dec 2022)
- Thread starter helen
- Start date
helen
Panic Sex Lady
In Afghanistan, the new campaign begins tomorrow, December 19, 2022.
View: https://twitter.com/PolioFreeAfghan/status/1602885503111634944?s=20
View: https://twitter.com/PolioFreeAfghan/status/1602885503111634944?s=20
helen
Panic Sex Lady
Mormon news
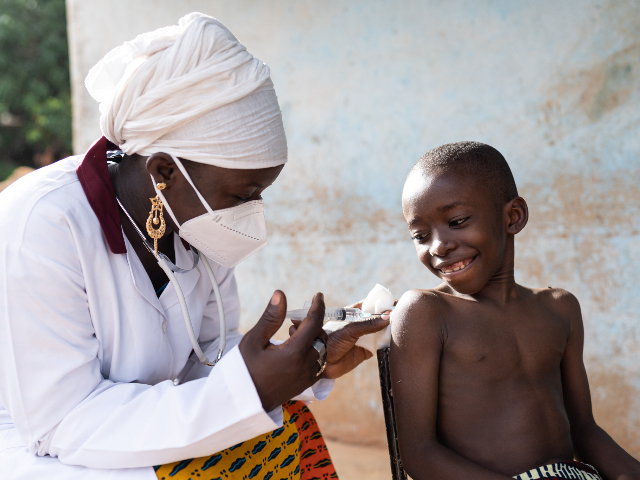
The polio donation ($5 million) goes to Rotary International, which has spent billions in recent decades to eradicate this paralyzing disease. Any donation to Rotary is matched 2-to-1 by the Bill & Melinda Gates Foundation — a financial blessing that presents an opportunity to eradicate the disease within our lifetime.
Michael K. McGovern, chair of Rotary’s International PolioPlus Committee, said, “Rotary is grateful for this very generous contribution from The Church of Jesus Christ of Latter-day Saints. The funding comes at a critical time for polio eradication efforts and will help protect children from lifelong paralysis due to the poliovirus.”
Half of the Church’s grant to Rotary will help African countries regain progress lost due to COVID-19. In under-immunized areas such as Malawi and Mozambique, wild poliovirus has reemerged. And communities in the Democratic Republic of the Congo and Nigeria are seeing polio outbreaks rarely seen in places where the disease was once eradicated.
 newsroom.churchofjesuschrist.org
newsroom.churchofjesuschrist.org
The polio donation ($5 million) goes to Rotary International, which has spent billions in recent decades to eradicate this paralyzing disease. Any donation to Rotary is matched 2-to-1 by the Bill & Melinda Gates Foundation — a financial blessing that presents an opportunity to eradicate the disease within our lifetime.
Michael K. McGovern, chair of Rotary’s International PolioPlus Committee, said, “Rotary is grateful for this very generous contribution from The Church of Jesus Christ of Latter-day Saints. The funding comes at a critical time for polio eradication efforts and will help protect children from lifelong paralysis due to the poliovirus.”
Half of the Church’s grant to Rotary will help African countries regain progress lost due to COVID-19. In under-immunized areas such as Malawi and Mozambique, wild poliovirus has reemerged. And communities in the Democratic Republic of the Congo and Nigeria are seeing polio outbreaks rarely seen in places where the disease was once eradicated.

Church of Jesus Christ Puts US$10 Million Toward Polio & Tetanus Vaccinations
The Church of Jesus Christ is giving US$10 million to help eradicate polio in Afghanistan and Pakistan, respond to outbreaks in Africa, and eliminate maternal and neonatal tetanus in 13 remaining countries throughout Af
helen
Panic Sex Lady
helen
Panic Sex Lady
Study Elizabeth Kenny's methods of treating polio patients. You might need to know.
Today’s Doodle celebrates Elizabeth Kenny, an Australian nurse who founded an alternative treatment for polio known as the Kenny Method.
Her exercises rehabilitated thousands of polio victims all over the world, and is considered one of the most effective forms of treatment prior to vaccines.
The Sister Kenny Memorial House, which celebrates her life’s work, opened on this day in Nobby, Queensland in 1997.
Kenny was born in 1880 in Warialda, New South Wales. She grew up in an impoverished farming community in rural Australia, where she received little formal education but was an avid reader who loved learning about medicine and human anatomy.
Although Kenny did not have the option to attend medical school, at 17 she forged her own path by volunteering at a hospital in Guyra. After shadowing nurses and doctors for more than a decade, Kenny gained enough working knowledge to open her own nursing practice in Darling Downs, Queensland.
In 1911, she encountered her first case of polio. She was unaware of the standard treatment at the time, which forced polio patients to lay in body casts for months which, in turn, caused muscle atrophy. This caused many polio victims to become permanently paralysed.
With her fresh perspective, Kenny realised the affected muscles were stiff, not permanently damaged. So she healed her patients by applying hot, wet compresses to the affected limbs, before having them perform gradual muscle strengthening exercises.
To the surprise of the medical community, her method worked! From then on, the exercises became known as the Kenny Method and news of this effective treatment spread far and wide.
Kenny traveled to America in the 1940s to open rehabilitation centers such as the Sister Kenny Institute in Minneapolis, which became a world-renowned center for polio treatment.
Her alternative method was so effective that she received honorary degrees from Rutgers University and the University of Rochester. President Franklin D. Roosevelt even invited her to lunch to discuss his own treatment.
Impressed by the number of polio victims that the Kenny Method rehabilitated, President Harry Truman authorised Kenny to enter the U.S. as she wished without a visa, a great honor only previously granted to one other non-US citizen.
g-elizabeth-kenny
Today’s Doodle celebrates Elizabeth Kenny, an Australian nurse who founded an alternative treatment for polio known as the Kenny Method.
Her exercises rehabilitated thousands of polio victims all over the world, and is considered one of the most effective forms of treatment prior to vaccines.
The Sister Kenny Memorial House, which celebrates her life’s work, opened on this day in Nobby, Queensland in 1997.
Kenny was born in 1880 in Warialda, New South Wales. She grew up in an impoverished farming community in rural Australia, where she received little formal education but was an avid reader who loved learning about medicine and human anatomy.
Although Kenny did not have the option to attend medical school, at 17 she forged her own path by volunteering at a hospital in Guyra. After shadowing nurses and doctors for more than a decade, Kenny gained enough working knowledge to open her own nursing practice in Darling Downs, Queensland.
In 1911, she encountered her first case of polio. She was unaware of the standard treatment at the time, which forced polio patients to lay in body casts for months which, in turn, caused muscle atrophy. This caused many polio victims to become permanently paralysed.
With her fresh perspective, Kenny realised the affected muscles were stiff, not permanently damaged. So she healed her patients by applying hot, wet compresses to the affected limbs, before having them perform gradual muscle strengthening exercises.
To the surprise of the medical community, her method worked! From then on, the exercises became known as the Kenny Method and news of this effective treatment spread far and wide.
Kenny traveled to America in the 1940s to open rehabilitation centers such as the Sister Kenny Institute in Minneapolis, which became a world-renowned center for polio treatment.
Her alternative method was so effective that she received honorary degrees from Rutgers University and the University of Rochester. President Franklin D. Roosevelt even invited her to lunch to discuss his own treatment.
Impressed by the number of polio victims that the Kenny Method rehabilitated, President Harry Truman authorised Kenny to enter the U.S. as she wished without a visa, a great honor only previously granted to one other non-US citizen.
g-elizabeth-kenny
helen
Panic Sex Lady
Elizabeth Kenny's method. You may need to know.
This is information they will control.
This is information they will control.
helen
Panic Sex Lady
Abstract
1. Forty-eight patients with infantile paralysis in 1941 have been treated by the methods of Sister Elizabeth Kenny of Australia, with encouraging and significant results when compared with twelve patients treated by older methods.
See table in the pdf file.
2. The presence of the symptoms of muscle spasm, mental alienation and incoordination in the acute stage of the disease has been verified.
3. The important rôle of untreated muscle spasm in forming muscle contractures and producing deformities has been demonstrated.
4. Treatment by means of bed rest in a natural position, hot moist packs, passive motion, and muscle re-education has proved successful in relieving these early symptoms when given as Sister Kenny has described.3
5. Immobilization by casts or splints is not necessary for the protection of weak muscles or to prevent deformities, and is actually harmful in prolonging muscle spasm and preventing the use of the Kenny routines.
6. Patients receiving the Kenny treatment are more comfortable, have better general health and nutrition, are more receptive to muscle training, have a superior morale, require a shorter period of bed rest and hospital care, and seem to have less residual paralysis and deformity than patients treated by older conventional methods. The Kenny treatment is the method of choice for the acute stage of infantile paralysis.

THE KENNY TREATMENT FOR INFANTILE PARALYSIS: A Comparison... : JBJS
een treated by the methods of Sister Elizabeth Kenny of Australia, with encouraging and significant results when compared with twelve patients treated by older methods. See table in the pdf file. 2. The presence of the symptoms of muscle spasm, mental alienation and incoordination in the acute...
helen
Panic Sex Lady
Skip Navigation

MENU
Home > Our Work > Tabletop Exercises
The Center hosts a series of tabletop exercises to illustrate the high-level strategic decisions and policies stakeholders will need to pursue to diminish the consequences of a severe pandemic. The training tabletop exercises are based on a fictional scenarios and inputs. These exercises are teaching and training resources for public health and government officials.
October 23, 2022
October 18, 2019
May 15, 2018
January 14, 2005
June 22-23, 2001

Our Mission
To protect people’s health from epidemics and disasters and ensure that communities are resilient to major challenges.
LINKS
Who We Are
Our Work
Resources
News
Contact
Donate
NEWSLETTERS
Community Health Monthly Roundup
COVID-19 Situation Reports
Health Security Headlines
Newsletter Sign Up
FELLOWSHIP
Emerging Leaders in Biosecurity (ELBI)
JOURNAL
Health Security
FOLLOW US ON:


JOHNS HOPKINS
CENTER FOR HEALTH SECURITY
621 E. Pratt Street
Suite 210
Baltimore, MD 21202
centerhealthsecurity@jhu.edu
Copyright © 2022 Johns Hopkins University | Web Policies | Site map | Site search

MENU
Home > Our Work > Tabletop Exercises
Tabletop Exercises
The Center hosts a series of tabletop exercises to illustrate the high-level strategic decisions and policies stakeholders will need to pursue to diminish the consequences of a severe pandemic.The Center hosts a series of tabletop exercises to illustrate the high-level strategic decisions and policies stakeholders will need to pursue to diminish the consequences of a severe pandemic. The training tabletop exercises are based on a fictional scenarios and inputs. These exercises are teaching and training resources for public health and government officials.
Catastrophic Contagion
The Johns Hopkins Center for Health Security, in partnership with WHO and the Bill & Melinda Gates Foundation, conducted Catastrophic Contagion, a pandemic tabletop exercise at the Grand Challenges Annual Meeting in Brussels, Belgium.October 23, 2022
Event 201
The Johns Hopkins Center for Health Security in partnership with the World Economic Forum and the Bill and Melinda Gates Foundation hosted Event 201, a high-level pandemic exercise on October 18, 2019, in New York, NY. The purpose of the exercise was to illustrate the pandemic preparedness efforts, response decisions, and cooperation required from global businesses, governments, and public health leaders that the world will need to diminish the large-scale economic and societal consequences of a severe pandemic.October 18, 2019
Clade X
The Center hosted a pandemic tabletop exercise in Washington, D.C., in May 2018. The goal of this exercise (“Clade X”) was to illustrate high-level strategic decisions and policies that the United States and the world will need to pursue in order to diminish the consequences of a severe pandemic. It addressed a pressing current concern, present plausible solutions, and be experientially engaging.May 15, 2018
Atlantic Storm
Atlantic Storm was a ministerial table-top exercise convened by the Center for Biosecurity, the Center for Transatlantic Relations of the Johns Hopkins University, and the Transatlantic Biosecurity Network. The exercise used a fictitious scenario designed to mimic a summit of transatlantic leaders forced to respond to a bioterrorist attack. The event website provides a comprehensive overview of the exercise and access to all materials and multimedia, including Atlantic Storm Interactive, and the after-action report, "Navigating the Storm."January 14, 2005
Dark Winter
The Dark Winter exercise portrayed a fictional scenario depicting a covert smallpox attack on US citizens. The scenario is set in 3 successive NSC meetings that take place over 2 weeks. The exercise was held at Andrews Air Force Base, Washington, DC. The Dark Winter script and other event materials are available through the conference web pages.June 22-23, 2001

Our Mission
To protect people’s health from epidemics and disasters and ensure that communities are resilient to major challenges.
LINKS
Who We Are
Our Work
Resources
News
Contact
Donate
NEWSLETTERS
Community Health Monthly Roundup
COVID-19 Situation Reports
Health Security Headlines
Newsletter Sign Up
FELLOWSHIP
Emerging Leaders in Biosecurity (ELBI)
JOURNAL
Health Security
FOLLOW US ON:


JOHNS HOPKINS
CENTER FOR HEALTH SECURITY
621 E. Pratt Street
Suite 210
Baltimore, MD 21202
centerhealthsecurity@jhu.edu
Copyright © 2022 Johns Hopkins University | Web Policies | Site map | Site search
Tristan
TB Fanatic
Monkeypox didn't go away. It just slowed down. It's mutating too. I thought another pandemic exercise was a joke, but it's not. My first response is to find it and find a way to fight it. But my oxygen levels are slipping into the 70's now, and the highs are getting lower. I'm finding stuff for you guys to study. Don't wait for 2025.
O2 sats in the 70's are dangerous... I sincerely hope you are receiving medical care!
helen
Panic Sex Lady
Polio Vaccines
Polio Vaccines — Precision Vaccinations
www.precisionvaccinations.com
Polio Vaccines Approved
There are two types of polio vaccines, an inactivated (killed) polio vaccine (IPV) was developed by Dr. Jonas Salk and first used in 1955. And a live attenuated (weakened) oral polio vaccine (OPV) developed by Dr. Albert Sabin and first used in 1961 reports the Global Polio Eradication Initiative (GPEI).
Polio Vaccine Availability
Since 2000, only the IPV vaccine has been available in the U.S. The U.S. Centers for Disease and Prevention (CDC) says the IPV can reduce how much poliovirus people shed, but it can't stop all virus transmission. In addition, the CDC's website stated on September 15, 2022, that it is unknown how long people who received IPV will be protected against polio.
The CDC's Advisory Committee on Immunization Practices (ACIP) reviewed poliovirus, polio vaccination, and polio epidemiology on October 19, 2022. The U.S. CDC published a poliovirus vaccine update on September 22, 2022, recommending children get four doses of any combination of IPV and trivalent oral polio vaccine (tOPV) or a primary series of at least three doses of IPV or tOPV.
The state of New York (NY) says adults who completed their polio vaccination but are at increased risk of contracting poliovirus may receive an IPV booster under specific situations. For polio vaccination recommendations for adults in NY, see Polio Vaccination: What Everyone Should Know.
The Imovax Polio®; IPOL® IPV vaccine is indicated for active immunization of infants (as young as six weeks), children, and adults to prevent poliomyelitis caused by poliovirus types 1, 2, and 3. The IPV protects against both wild-type polio and this weakened poliovirus strain. For details and age groups, refer to the ACIP IPV catch-up vaccine table.
Kinrix is indicated for active immunization against diphtheria, tetanus, pertussis, and poliomyelitis as the fifth dose in the diphtheria, tetanus, and acellular pertussis (DTaP) vaccine series and the fourth dose in the IPV series in children 4 through 6 years of age whose previous DTaP vaccine doses have been with Infanrix and/or Pediarix for the first three doses and Infanrix for the fourth dose.
Pediarix is a vaccine for active immunization against diphtheria, tetanus, pertussis, and infection caused by all known subtypes of hepatitis B virus and poliomyelitis.
Pentacel is a multi-vaccine consisting of Diphtheria, Tetanus Toxoids, and Acellular Pertussis Adsorbed and Inactivated Poliovirus (DTaP-IPV) component as well as an ActHIB® vaccine component.
Quadracel vaccine is indicated for active immunization against diphtheria, tetanus, pertussis, and poliomyelitis.
Sabin IPV (sIPV) inactivated vaccine produced by SINOVAC Biotech Ltd. is indicated to prevent the Wild Polio Virus and was WHO prequalified in June 2022.
Oral Polio Vaccine
Oral polio vaccines (OPV) contain a weakened strain of the poliovirus that has changed over time and now behaves similarly to a wild-type polio infection. In addition, OPV can lead to vaccine-derived poliovirus, which can enter the blood and paralyze an unvaccinated person and is not offered in the U.S.
In 2016, the CDC announced to address the risks posed by type 2 circulating vaccine-derived polioviruses, which have caused hundreds of paralytic poliomyelitis cases since 2006, the type 2 component of oral poliovirus vaccine was withdrawn through a switch from trivalent oral poliovirus vaccine (tOPV) to bivalent oral poliovirus vaccine (bOPV), which contains only attenuated viruses of types 1 and 3. However, the bOPV vaccine does not give immunity against serotype 2. This change reduced the risk of tOPV seeding new type 2 circulating vaccine-derived polioviruses (cVDPV2).
As of December 30, 2022, the tOPV remains in use with children in countries such as Somalia. In addition, OPVs are inexpensive (U.S. $0.12) for countries procuring vaccines through UNICEF.
Novel Oral Polio Vaccine Type 2 (nOPV2)
In November 2020, the nOPV2 vaccine, a modified version of the type 2 monovalent OPV (mOPV2) vaccine, was recommended under WHO's Emergency Use Listing procedure. The first-in-human clinical trial was conducted in 2017 at the University of Antwerp and found nOPV2 safe and efficacious. The Lancet published these findings in June 2019. As of December 2022, approximately 550 million doses of nOPV2 have been administered in about 25 countries. Following a review of the nOPV2, the WHO's Strategic Advisory Group of Experts on immunization (SAGE) endorsed a vaccine transition program on October 11, 2021.
Polio Vaccine Price
The U.S. CDC confirmed the Vaccines For Children program is a federally funded program that provides IPV vaccines and medicines at no cost to children to children who might not otherwise be vaccinated because of their inability to pay. For people in the U.S. not covered by health insurance, a polio booster typically costs about $100. So, for example, pharmacies may charge about $100 for an IPV polio booster shot.
The U.S. CDC Vaccine Price List was updated in September 2022. This UNICEF table provides an overview of Oral polio vaccine prices contracted with suppliers. And additional polio vaccine prices and discount information are available at InstantRx™.
Poliovirus Outbreaks 2022
On November 30, 2022, the U.S. CDC reissued a Global Polio Level 2 Travel Alert. For the latest news regarding poliovirus detections and polio cases, visit this PrecisionVaccinations webpage.
Polio Vaccine News 2022
November 8, 2022 - PharmaJet® announced that their PharmaJet Tropis® Needle-free Injection System (NFIS) would be used in a door-to-door children immunization campaign to reduce the outbreak of cMPV2. The initial pilot was in the Sokoto North local government area of Nigeria.
November 1, 2022 - The WHO announced its vaccine Committee unanimously agreed that the risk of the international spread of poliovirus remains a Public Health Emergency of International Concern and recommended the extension of Temporary Recommendations for a further three months.
October 24, 2022 - PharmaJet® announced they received a multi-year, $1.5 million grant from the United States Agency for International Development to evaluate the impact of intradermal (I.D.) vaccine administration using their Tropis® Needle-free Injection System. The project will measure vaccine coverage and cost using Tropis ID for fractional inactivated poliovirus vaccine delivery compared to standard intramuscular delivery using a needle and syringe.
August 6, 1955 - The JAMA Network published - EVALUATION OF THE 1954 POLIOMYELITIS VACCINE FIELD TRIAL: 516 total cases of paralytic polio accumulated in the 1954 polio field trial, and vaccination reduced its incidence by 71.1% and 62.4%, relative to the placebo and observational-control groups, respectively.
Polio Vaccines — Precision Vaccinations
www.precisionvaccinations.com
Polio Vaccines Approved
There are two types of polio vaccines, an inactivated (killed) polio vaccine (IPV) was developed by Dr. Jonas Salk and first used in 1955. And a live attenuated (weakened) oral polio vaccine (OPV) developed by Dr. Albert Sabin and first used in 1961 reports the Global Polio Eradication Initiative (GPEI).
Polio Vaccine Availability
Since 2000, only the IPV vaccine has been available in the U.S. The U.S. Centers for Disease and Prevention (CDC) says the IPV can reduce how much poliovirus people shed, but it can't stop all virus transmission. In addition, the CDC's website stated on September 15, 2022, that it is unknown how long people who received IPV will be protected against polio.
The CDC's Advisory Committee on Immunization Practices (ACIP) reviewed poliovirus, polio vaccination, and polio epidemiology on October 19, 2022. The U.S. CDC published a poliovirus vaccine update on September 22, 2022, recommending children get four doses of any combination of IPV and trivalent oral polio vaccine (tOPV) or a primary series of at least three doses of IPV or tOPV.
The state of New York (NY) says adults who completed their polio vaccination but are at increased risk of contracting poliovirus may receive an IPV booster under specific situations. For polio vaccination recommendations for adults in NY, see Polio Vaccination: What Everyone Should Know.
The Imovax Polio®; IPOL® IPV vaccine is indicated for active immunization of infants (as young as six weeks), children, and adults to prevent poliomyelitis caused by poliovirus types 1, 2, and 3. The IPV protects against both wild-type polio and this weakened poliovirus strain. For details and age groups, refer to the ACIP IPV catch-up vaccine table.
Kinrix is indicated for active immunization against diphtheria, tetanus, pertussis, and poliomyelitis as the fifth dose in the diphtheria, tetanus, and acellular pertussis (DTaP) vaccine series and the fourth dose in the IPV series in children 4 through 6 years of age whose previous DTaP vaccine doses have been with Infanrix and/or Pediarix for the first three doses and Infanrix for the fourth dose.
Pediarix is a vaccine for active immunization against diphtheria, tetanus, pertussis, and infection caused by all known subtypes of hepatitis B virus and poliomyelitis.
Pentacel is a multi-vaccine consisting of Diphtheria, Tetanus Toxoids, and Acellular Pertussis Adsorbed and Inactivated Poliovirus (DTaP-IPV) component as well as an ActHIB® vaccine component.
Quadracel vaccine is indicated for active immunization against diphtheria, tetanus, pertussis, and poliomyelitis.
Sabin IPV (sIPV) inactivated vaccine produced by SINOVAC Biotech Ltd. is indicated to prevent the Wild Polio Virus and was WHO prequalified in June 2022.
Oral Polio Vaccine
Oral polio vaccines (OPV) contain a weakened strain of the poliovirus that has changed over time and now behaves similarly to a wild-type polio infection. In addition, OPV can lead to vaccine-derived poliovirus, which can enter the blood and paralyze an unvaccinated person and is not offered in the U.S.
In 2016, the CDC announced to address the risks posed by type 2 circulating vaccine-derived polioviruses, which have caused hundreds of paralytic poliomyelitis cases since 2006, the type 2 component of oral poliovirus vaccine was withdrawn through a switch from trivalent oral poliovirus vaccine (tOPV) to bivalent oral poliovirus vaccine (bOPV), which contains only attenuated viruses of types 1 and 3. However, the bOPV vaccine does not give immunity against serotype 2. This change reduced the risk of tOPV seeding new type 2 circulating vaccine-derived polioviruses (cVDPV2).
As of December 30, 2022, the tOPV remains in use with children in countries such as Somalia. In addition, OPVs are inexpensive (U.S. $0.12) for countries procuring vaccines through UNICEF.
Novel Oral Polio Vaccine Type 2 (nOPV2)
In November 2020, the nOPV2 vaccine, a modified version of the type 2 monovalent OPV (mOPV2) vaccine, was recommended under WHO's Emergency Use Listing procedure. The first-in-human clinical trial was conducted in 2017 at the University of Antwerp and found nOPV2 safe and efficacious. The Lancet published these findings in June 2019. As of December 2022, approximately 550 million doses of nOPV2 have been administered in about 25 countries. Following a review of the nOPV2, the WHO's Strategic Advisory Group of Experts on immunization (SAGE) endorsed a vaccine transition program on October 11, 2021.
Polio Vaccine Price
The U.S. CDC confirmed the Vaccines For Children program is a federally funded program that provides IPV vaccines and medicines at no cost to children to children who might not otherwise be vaccinated because of their inability to pay. For people in the U.S. not covered by health insurance, a polio booster typically costs about $100. So, for example, pharmacies may charge about $100 for an IPV polio booster shot.
The U.S. CDC Vaccine Price List was updated in September 2022. This UNICEF table provides an overview of Oral polio vaccine prices contracted with suppliers. And additional polio vaccine prices and discount information are available at InstantRx™.
Poliovirus Outbreaks 2022
On November 30, 2022, the U.S. CDC reissued a Global Polio Level 2 Travel Alert. For the latest news regarding poliovirus detections and polio cases, visit this PrecisionVaccinations webpage.
Polio Vaccine News 2022
November 8, 2022 - PharmaJet® announced that their PharmaJet Tropis® Needle-free Injection System (NFIS) would be used in a door-to-door children immunization campaign to reduce the outbreak of cMPV2. The initial pilot was in the Sokoto North local government area of Nigeria.
November 1, 2022 - The WHO announced its vaccine Committee unanimously agreed that the risk of the international spread of poliovirus remains a Public Health Emergency of International Concern and recommended the extension of Temporary Recommendations for a further three months.
October 24, 2022 - PharmaJet® announced they received a multi-year, $1.5 million grant from the United States Agency for International Development to evaluate the impact of intradermal (I.D.) vaccine administration using their Tropis® Needle-free Injection System. The project will measure vaccine coverage and cost using Tropis ID for fractional inactivated poliovirus vaccine delivery compared to standard intramuscular delivery using a needle and syringe.
August 6, 1955 - The JAMA Network published - EVALUATION OF THE 1954 POLIOMYELITIS VACCINE FIELD TRIAL: 516 total cases of paralytic polio accumulated in the 1954 polio field trial, and vaccination reduced its incidence by 71.1% and 62.4%, relative to the placebo and observational-control groups, respectively.
helen
Panic Sex Lady
Money, money, money, money.
Polio Vaccine to Witness Huge Growth by 2031 | GSK, Sanofi, Bibcol, Serum Institute, Tiantan Biological, IMBCA, Panacea Biotec Ltd, Bio-Med, Halfkin Bio-Pharmaceuticals
Polio Vaccine to Witness Huge Growth by 2031 | GSK, Sanofi, Bibcol, Serum Institute, Tiantan Biological, IMBCA, Panacea Biotec Ltd, Bio-Med, Halfkin Bio-Pharmaceuticals - LatAm Insurance Review
The proposed Polio Vaccine Market report will encompass all the qualitative & quantitative aspec
www.insurancelatam.com
helen
Panic Sex Lady
Indonesia Deploys Novel Polio Vaccines for Children
According to the World Health Organization (WHO), the Republic of Indonesia recently joined an expanding list of countries deploying a new polio vaccine to curtail an active outbreak. The WHO announced on December 19, 2022, Indonesia’s Ministry of Health had notified the WHO of a confirmed case...

Egypt vaccinates over 3.2 million children in latest polio vaccination campaign
Egypt’s Ministry of Health announced vaccinating more than 3.2 million children, who ages five years or less, during the latest limited vaccination campaign in Cairo and Giza earlier this month.
Pakistan

All Set To Kick Start National Immunization Drive Against Polio: EOC - UrduPoint
The first National Immunization Drive (NID) against polio to kick start in two phases in the province with the first phase starting from January 2, 2023, in Bannu and DI Khan divisions wherein 1.08 million children below 5 years would be administered polio vaccine and Vitamin A supplements. This...

Mozambique vaccinates 7 mn children against polio
Mozambique vaccinates 7 mn children against polio
 in.investing.com
in.investing.com
helen
Panic Sex Lady
New York
County To Accept Polio Vaccine | News, Sports, Jobs
www.post-journal.com
Dr. Michael Faulk, interim public health director, speaks to the Chautauqua County Legislature. Photo by Gregory Bacon
Chautauqua County is moving ahead to help get people vaccinated against polio for those who want it.
During last week’s county Legislature meeting, lawmakers voted unanimously to accept Poliovirus vaccine from the state Health Department.
There is no charge for the vaccine. In the past the county has accepted COVID-19 vaccine as well as vaccine for monkeypox.
Dr. Michael Faulk, interim public health director, discussed the program at both the full legislature meeting as well as during committee meetings from the week prior.
“Several months ago Rockland County in New York state reported a positive polio case in a young individual that resulted in significant neurologic deficits, which polio does, essentially causing paralysis,” he said.
There have been no other positive cases reported in Chautauqua County or elsewhere in the state.
Faulk said those at risk for polio are the unvaccinated.
“The state is basically saying we want to have the opportunity for those that are willing, to get vaccinated, to have the vaccine ready and available at no cost,” he said.
Faulk said there are several people in the county that are unvaccinated but have changed their mind about the polio vaccine. “We want to be there for those who decide that that’s important to them,” he said.
Faulk noted the Amish population in particular has a large number of people who are unvaccinated.
“Certain sects of the Amish are open to the idea of vaccination,” he said, noting the county just recently held a vaccination clinic in December in an Amish community.
Faulk calls polio “a devastating illness” and wants to help people understand the benefits of being vaccinated. “The last thing we want is to see polio rear is ugly head in our society. It’s a horrible, terrible disease, causing terrible neurologic deficits that last a lifetime,” he said.
Faulk sees the Public Health Department’s role to help educate people about these types of diseases and offer vaccines for those who want it. “It’s a personal decision that they make between themselves and their physicians or us as the Health Department. We’ll give them as much information as we can, so that they can make an informed decision on whether or not it’s right for them,” he said.
The resolution accepting the polio vaccine from the state was unanimously approved.
Newsletter
Today's breaking news and more in your inbox
County To Accept Polio Vaccine | News, Sports, Jobs
www.post-journal.com
Dr. Michael Faulk, interim public health director, speaks to the Chautauqua County Legislature. Photo by Gregory Bacon
Chautauqua County is moving ahead to help get people vaccinated against polio for those who want it.
During last week’s county Legislature meeting, lawmakers voted unanimously to accept Poliovirus vaccine from the state Health Department.
There is no charge for the vaccine. In the past the county has accepted COVID-19 vaccine as well as vaccine for monkeypox.
Dr. Michael Faulk, interim public health director, discussed the program at both the full legislature meeting as well as during committee meetings from the week prior.
“Several months ago Rockland County in New York state reported a positive polio case in a young individual that resulted in significant neurologic deficits, which polio does, essentially causing paralysis,” he said.
There have been no other positive cases reported in Chautauqua County or elsewhere in the state.
Faulk said those at risk for polio are the unvaccinated.
“The state is basically saying we want to have the opportunity for those that are willing, to get vaccinated, to have the vaccine ready and available at no cost,” he said.
Faulk said there are several people in the county that are unvaccinated but have changed their mind about the polio vaccine. “We want to be there for those who decide that that’s important to them,” he said.
Faulk noted the Amish population in particular has a large number of people who are unvaccinated.
“Certain sects of the Amish are open to the idea of vaccination,” he said, noting the county just recently held a vaccination clinic in December in an Amish community.
Faulk calls polio “a devastating illness” and wants to help people understand the benefits of being vaccinated. “The last thing we want is to see polio rear is ugly head in our society. It’s a horrible, terrible disease, causing terrible neurologic deficits that last a lifetime,” he said.
Faulk sees the Public Health Department’s role to help educate people about these types of diseases and offer vaccines for those who want it. “It’s a personal decision that they make between themselves and their physicians or us as the Health Department. We’ll give them as much information as we can, so that they can make an informed decision on whether or not it’s right for them,” he said.
The resolution accepting the polio vaccine from the state was unanimously approved.
Newsletter
Today's breaking news and more in your inbox
helen
Panic Sex Lady
The new oral polio vaccine will get CDC emergency use authorization because there is a growing shortage of syringes.
No syringe = no IPV (inactivated polio virus) injected vaccines
The syringe shortage, explained | PATH
www.path.org
A: Right now, all countries donating COVID-19 vaccines—whether through mechanisms like COVAX or directly to other countries—should work in tandem with global procurement agencies like UNICEF to bundle their doses together with essential equipment like the appropriate syringes. Without the appropriate syringes, these vaccines could sit unused or expire waiting for the correct syringes to arrive.
At PATH, we’re working to close the gap in several ways. We are:
No syringe = no IPV (inactivated polio virus) injected vaccines
The syringe shortage, explained | PATH
www.path.org
A: Right now, all countries donating COVID-19 vaccines—whether through mechanisms like COVAX or directly to other countries—should work in tandem with global procurement agencies like UNICEF to bundle their doses together with essential equipment like the appropriate syringes. Without the appropriate syringes, these vaccines could sit unused or expire waiting for the correct syringes to arrive.
At PATH, we’re working to close the gap in several ways. We are:
- Assessing the market for a different type of syringe called “reuse prevention”. This syringe has safety features that are similar to an AD syringe, but it is not fixed dose and thus requires measurement of the dose by the person administering the vaccine. In addition to understanding the market, we are gathering data on the countries in which these syringes may be suitable for use.
- Partnering with UNICEF and other global stakeholders to characterize supply chain challenges and develop strategies to help ensure that syringes and vaccines arrive together. This may include prepositioning supply as it is produced and revising procurement mechanisms.
- Working to bolster supply capacity, particularly for manufacturers operating in LMICs. At present, there are only two AD syringe suppliers on the African continent, so bringing in additional local production capacity is important, such as the recent investment by the Bill & Melinda Gates Foundation in a Kenyan AD syringe manufacturer.
- For health workers, it would simplify the selection of the correct syringe to deliver a vaccine dose and help ensure the right-sized AD syringe is available when and where it is needed.
- For manufacturers, it would avoid the need to invest in production equipment for new syringe sizes and maximize efficiency by avoiding the need to switch lines back and forth between production of different-sized AD syringes.
- For procurement agencies, it would help streamline advanced procurement and distribution of AD syringes, as vaccine shipments would not need to be matched with the corresponding syringe type.
helen
Panic Sex Lady
Meanwhile, 13 months ago...
COVID-19 pandemic brings global syringe shortage into sharp focus
news.un.org
Based on a scenario where around seven billion people need two doses of coronavirus vaccine between now and 2023, the UN health agency said that a shortage of at least one billion syringes “could occur”, if manufacturing does not pick up.
Lisa Hedman, WHO Senior Advisor, from the Access to Medicines and Health Products division, warned that a generation of children might miss scheduled immunization jabs unless manufacturers find a way to make more single-use disposable syringes.
She told journalists in Geneva that more than 6.8 billion doses of COVID-19 vaccines are being administered globally per year, which is nearly double the number of routine inoculations delivered annually:
“A shortage of syringes is unfortunately a real possibility and here’s some more numbers. That the global manufacturing capacity of around six billion a year for immunization syringes it’s pretty clear that a deficit in 2022 of over a billion could happen if we continue with business as usual.”
Ms. Hedman explained that reusing syringes even after they have been sterilized was not advised, as harmful bacteria remained present.
She also noted that syringes were particularly prone to transport delays because they took up 10 times the space of a vaccine.
At the meeting, according to a press release, all participants agreed on the urgency of delivering more vaccine doses to low-income countries, where less than 2.5 per cent of the population has been fully vaccinated.
The objective of the meeting was to identify how to ensure more equitable distribution of vaccines and all those participating pledged to continue working together to gain greater clarity on donations, vaccine swaps and delivery schedules, so that distribution of the life-saving vaccines can be more effectively targeted towards those countries most in need.
The meeting of the Multilateral Leaders Task Force on COVID-19 built on technical work undertaken by multidisciplinary teams during the months of September and October.
During the consultations, the heads of the four organizations and the CEOs also examined how best to tackle trade-related bottlenecks; how to improve the donation process; what additional steps are needed to reach the vaccination target of 40 per cent of people in all countries by the end of the year; and how to improve transparency and data sharing with the IMF-WHO Vaccine Supply Forecast Dashboard and the Multilateral Leaders Task Force.
The effort will require close collaboration between manufacturers, governments and the international COVAX initiative, on enhanced delivery schedules, especially for doses that are being donated.
COVID-19 pandemic brings global syringe shortage into sharp focus
news.un.org
Based on a scenario where around seven billion people need two doses of coronavirus vaccine between now and 2023, the UN health agency said that a shortage of at least one billion syringes “could occur”, if manufacturing does not pick up.
Lisa Hedman, WHO Senior Advisor, from the Access to Medicines and Health Products division, warned that a generation of children might miss scheduled immunization jabs unless manufacturers find a way to make more single-use disposable syringes.
No place for shortcuts
“When you think about the magnitude of the number of injections being given to respond to the pandemic, this is not a place where we can afford shortcuts, shortages or anything short of full safety for patients and healthcare staff,” the WHO expert said.She told journalists in Geneva that more than 6.8 billion doses of COVID-19 vaccines are being administered globally per year, which is nearly double the number of routine inoculations delivered annually:
“A shortage of syringes is unfortunately a real possibility and here’s some more numbers. That the global manufacturing capacity of around six billion a year for immunization syringes it’s pretty clear that a deficit in 2022 of over a billion could happen if we continue with business as usual.”
Ms. Hedman explained that reusing syringes even after they have been sterilized was not advised, as harmful bacteria remained present.
She also noted that syringes were particularly prone to transport delays because they took up 10 times the space of a vaccine.
Urgent need to ramp up shots
Meanwhile, the heads of the International Monetary Fund (IMF), World Bank Group, WHO and the World Trade Organization (WTO) held a follow up session of High-Level Consultations with the CEOs of leading COVID-19 vaccine manufacturing companies on Tuesday.At the meeting, according to a press release, all participants agreed on the urgency of delivering more vaccine doses to low-income countries, where less than 2.5 per cent of the population has been fully vaccinated.
The objective of the meeting was to identify how to ensure more equitable distribution of vaccines and all those participating pledged to continue working together to gain greater clarity on donations, vaccine swaps and delivery schedules, so that distribution of the life-saving vaccines can be more effectively targeted towards those countries most in need.
The meeting of the Multilateral Leaders Task Force on COVID-19 built on technical work undertaken by multidisciplinary teams during the months of September and October.
During the consultations, the heads of the four organizations and the CEOs also examined how best to tackle trade-related bottlenecks; how to improve the donation process; what additional steps are needed to reach the vaccination target of 40 per cent of people in all countries by the end of the year; and how to improve transparency and data sharing with the IMF-WHO Vaccine Supply Forecast Dashboard and the Multilateral Leaders Task Force.
The effort will require close collaboration between manufacturers, governments and the international COVAX initiative, on enhanced delivery schedules, especially for doses that are being donated.
helen
Panic Sex Lady
Where syringes are manufactured
Who are the world's biggest needle and syringe manufacturers?
www.nsmedicaldevices.com
...The company posted an annual revenue of almost $1.15bn for 2019, and employs 7,770 people in more than 30 countries – with manufacturing concentrated in the US, UK, Mexico, Italy, Germany, Czech Republic, and China.
Who are the world's biggest needle and syringe manufacturers?
www.nsmedicaldevices.com
...The company posted an annual revenue of almost $1.15bn for 2019, and employs 7,770 people in more than 30 countries – with manufacturing concentrated in the US, UK, Mexico, Italy, Germany, Czech Republic, and China.
psychgirl
TB Fanatic
Yes, thankfully not in my area “yet”… I flat out asked my practitioner if she was seeing any antibiotic shortages and she said no…I got mine filled literally within 30 minutes of her calling it in last week, thank God.You know how you can't get good help these days? COVID-19 and flu and RSV and strep are wiping out workers.
helen
Panic Sex Lady
Get the young people in the U.S. fully vaccinated with inactive polio virus IPV while you still can.
Global Circulating Vaccine-derived Poliovirus (cVDPV) as of 270 December 2022
If a population is seriously under-immunized, there are enough susceptible children for the excreted vaccine-derived polioviruses to begin circulating in the community. If the vaccine-virus is able to circulate for a prolonged period of time uninterrupted, it can mutate and, over the course of 12-18 months, reacquire neurovirulence. These viruses are called circulating vaccine-derived polioviruses (cVDPV).
The lower the population immunity, the longer these viruses survive. The longer they survive, the more they replicate, change, and exchange genetic material with other enteroviruses as they spread through a community.
If a population is fully immunized against polio, it will be protected against the spread of both wild and vaccine strains of poliovirus.

 polioeradication.org
polioeradication.org
Global Circulating Vaccine-derived Poliovirus (cVDPV) as of 270 December 2022
If a population is seriously under-immunized, there are enough susceptible children for the excreted vaccine-derived polioviruses to begin circulating in the community. If the vaccine-virus is able to circulate for a prolonged period of time uninterrupted, it can mutate and, over the course of 12-18 months, reacquire neurovirulence. These viruses are called circulating vaccine-derived polioviruses (cVDPV).
The lower the population immunity, the longer these viruses survive. The longer they survive, the more they replicate, change, and exchange genetic material with other enteroviruses as they spread through a community.
If a population is fully immunized against polio, it will be protected against the spread of both wild and vaccine strains of poliovirus.

GPEI-Circulating vaccine-derived poliovirus
helen
Panic Sex Lady
You don't have to think up a weird mutation of polio to turn it into "enterovirius respiratory syndrome" if it occurs with a massive spike of covid, flu, or RSV coughing and sneezing.
Mask up.
Poliovirus is very contagious and spreads through person-to-person contact. It spreads through contact with the stool (poop) of an infected person or droplets from a sneeze or cough. If you get stool or droplets from an infected person on your hands and you touch your mouth, you can get infected.Aug 24, 2022

CDC Works 24/7 › what-is-polio
Mask up.
Poliovirus is very contagious and spreads through person-to-person contact. It spreads through contact with the stool (poop) of an infected person or droplets from a sneeze or cough. If you get stool or droplets from an infected person on your hands and you touch your mouth, you can get infected.Aug 24, 2022
CDC Works 24/7 › what-is-polio
Polio Frequently Asked Questions (FAQ) - CDC
helen
Panic Sex Lady
September 2022
Chemical shortages. Water treatment. Polio spreads in contaminated water. Other diseases do too, but this thread is about Catastrophic Contagion and polio.
Residents in Jackson, Mississippi told to keep their mouths closed in shower amid water crisis
www.nydailynews.com
The water crisis in Mississippi’s capital city has become so dire that residents are being told to shower with cold water — and to keep their mouths shut while they do it.
The director of health protection for the Mississippi State Department of Health, Jim Craig, issued the recommendation on Wednesday for residents in Jackson, who have been living with little to no water flowing from their faucets for the last four days. He said that while it is still safe to bathe in the water, people should not “open your mouth while you’re in the shower.”
Chemical shortages. Water treatment. Polio spreads in contaminated water. Other diseases do too, but this thread is about Catastrophic Contagion and polio.
Residents in Jackson, Mississippi told to keep their mouths closed in shower amid water crisis
www.nydailynews.com
The water crisis in Mississippi’s capital city has become so dire that residents are being told to shower with cold water — and to keep their mouths shut while they do it.
The director of health protection for the Mississippi State Department of Health, Jim Craig, issued the recommendation on Wednesday for residents in Jackson, who have been living with little to no water flowing from their faucets for the last four days. He said that while it is still safe to bathe in the water, people should not “open your mouth while you’re in the shower.”
magnetic1
Veteran Member
You need oxygen. I hope you are getting help! Prayers sentO2 sats in the 70's are dangerous... I sincerely hope you are receiving medical care!
psychgirl
TB Fanatic
Make sure if around others, for whatever reason, hopefully they flush the toilet with the lid down!You don't have to think up a weird mutation of polio to turn it into "enterovirius respiratory syndrome" if it occurs with a massive spike of covid, flu, or RSV coughing and sneezing.
Mask up.
Poliovirus is very contagious and spreads through person-to-person contact. It spreads through contact with the stool (poop) of an infected person or droplets from a sneeze or cough. If you get stool or droplets from an infected person on your hands and you touch your mouth, you can get infected.Aug 24, 2022
View attachment 388107
CDC Works 24/7 › what-is-polio
Polio Frequently Asked Questions (FAQ) - CDC
The “spray” in public bathrooms may spread it, idk?
L.A.B.
Goodness before greatness.
You need oxygen. I hope you are getting help! Prayers sent
WTH is happening that oxygen saturation is spiraling so low?
helen
Panic Sex Lady
It's odd. The low numbers in the last five days are 79, 84, 86, 87, and 91, but the highs are higher. 96, 97, 98, 99, 99. I get outside and work more since the days are getting longer. Maybe the wood stove is sucking the oxygen too low at night.WTH is happening that oxygen saturation is spiraling so low?
magnetic1
Veteran Member
Are the lows during the night? I just went through that and have to use a little supplemental oxygen while asleep. Maybe that is it? Please check with Dr as they can do an easy overnight home test.It's odd. The low numbers in the last five days are 79, 84, 86, 87, and 91, but the highs are higher. 96, 97, 98, 99, 99. I get outside and work more since the days are getting longer. Maybe the wood stove is sucking the oxygen too low at night.
helen
Panic Sex Lady
Doctors are swamped. Labs are perfect. Blood pressure is perfect. No apnea. But there is a wood stove and sleep near it. Some of the highs while awake are in the 80's. I weatherize maybe too much.Are the lows during the night? I just went through that and have to use a little supplemental oxygen while asleep. Maybe that is it? Please check with Dr as they can do an easy overnight home test.
magnetic1
Veteran Member
That sounds like what I have going on. Erratic so now have to have oxygen at night so no major lows anymore and during the day I am to watch levels and use if need be. It’s a PITA.Doctors are swamped. Labs are perfect. Blood pressure is perfect. No apnea. But there is a wood stove and sleep near it. Some of the highs while awake are in the 80's. I weatherize maybe too much.
You are not taking metoprolol by any chance?
helen
Panic Sex Lady
No meds ever until shingles meds this week.That sounds like what I have going on. Erratic so now have to have oxygen at night so no major lows anymore and during the day I am to watch levels and use if need be. It’s a PITA.
You are not taking metoprolol by any chance?
magnetic1
Veteran Member
So can’t be a meds issue, then. Allergies or asthma set off by the wood stove? Good luck figuring this out!No meds ever until shingles meds this week.
helen
Panic Sex Lady
I'm vax plus C-19*3. Shingles increases heart attack and stroke by 75% for the next year. I'm dead.So can’t be a meds issue, then. Allergies or asthma set off by the wood stove? Good luck figuring this out!
magnetic1
Veteran Member
Not yet! Do you take the FLCCNET treatment? You are too tough to just lay down and roll over.I'm vax plus C-19*3. Shingles increases heart attack and stroke by 75% for the next year. I'm dead.
helen
Panic Sex Lady
I've got the full monty. Since it's 2023, I guess I'll start it up again. Aren't we all tired of this? And the OP, that's just more coming.Not yet! Do you take the FLCCNET treatment? You are too tough to just lay down and roll over.
But ivermectin might work on enterovirus.


magnetic1
Veteran Member
Atta girl!I've got the full monty. Since it's 2023, I guess I'll start it up again. Aren't we all tired of this? And the OP, that's just more coming.
But ivermectin might work on enterovirus.

bw
Fringe Ranger
I'm not ready for the Panic Sex Lady to exit my life. Don't do that.I'm dead.
helen
Panic Sex Lady
Panic Sex Lady rises to her patriotic duty as long as she can get a rise out of one man...I'm not ready for the Panic Sex Lady to exit my life. Don't do that.
psychgirl
TB Fanatic
Stop it!!I'm vax plus C-19*3. Shingles increases heart attack and stroke by 75% for the next year. I'm dead.
Immediately!!

