Heliobas Disciple
TB Fanatic
NAC & COVID
Examining some of the evidence for NAC use in treating and preventing COVID.
NAC & COVID
Examining some of the evidence for NAC use in treating and preventing COVID.
Modern Discontent
Aug 17
So far we’ve gone over some of the pharmacological effects of NAC and pointing to its ability to restore glutathione levels being its main mechanism of action.
Here, we’ll look at some of the clinical data in regards to NAC use against COVID and see what the evidence tells us.
A few important studies
Before we look specifically at COVID, I wanted to cite two studies that have been referenced quite a few times in the literature. These two studies are likely what was used to make a case for the use of NAC, similar to other repurposed drugs such as Ivermectin or Hydroxychloroquine which relied on previous clinical studies.
Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment (De Flora, et. al.)
One such study that has been referenced multiple times is a study from De Flora, et. al. which looked at long-term NAC supplementation and whether that would attenuate symptoms of the flu during the cold season.
In this study, researchers provided effervescent NAC tablets1 to participants and measured their rate of influenza-like infection over the cold seasons. They also measured participants' cell-mediated immunity to see if NAC would have an effect on immunological responses to infections:
The present multicentric study, referred to as the NACIS study (acronym for N-acetylcysteine in Immune System), was designed in order to evaluate the efficacy of NAC, administered orally throughout the cold season, in preventing the occurrence and reducing the severity of influenza-like episodes in general, and specifically those caused by the influenza A/H1N1 virus. Another goal of this study was to assess the effect of NAC treatment on cell-mediated immunity.
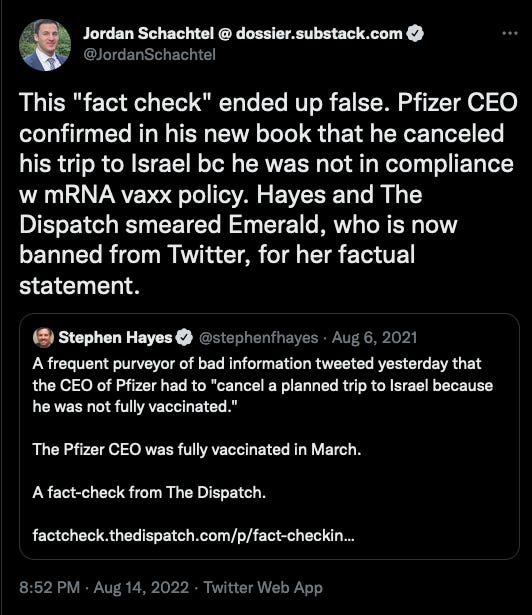
The researchers found that the NAC group had fewer incidences of influenza-like symptoms and that symptoms were more mild within the NAC group compared to the placebo group:
Of the 99 influenza-like episodes occurring in 62 subjects within the placebo group, 47 (48%) were classified as mild, 46 (47%) as moderate, and 6 (6%) as severe, according to the criteria reported in "Materials and methods". Of the 46 influenza-like episodes occurring in 37 subjects within the NAC group, 33 (72%) were classified as mild, 12 (26%) as moderate, and 1 (2%) as severe. As assessed by χ2 analysis, the frequency of mild forms was higher in the NAC group than in the placebo group, with a difference approaching the significance threshold (p=0.06), whereas the frequency of moderate forms was significantly higher in the placebo group than in the NAC group (p=0.02). The differences were particularly pronounced by mid-season, after 3 months of treatment, when mild, moderate and severe episodes were 9, 22 and 3 in the placebo group and 8, 4 and 0 in the NAC group, respectively (p=0.002), and after 4 months, when mild, moderate and severe episodes were 10, 6 and 0 in the placebo group and 3, 0 and 0 in the NAC group, respectively (p=0.006).
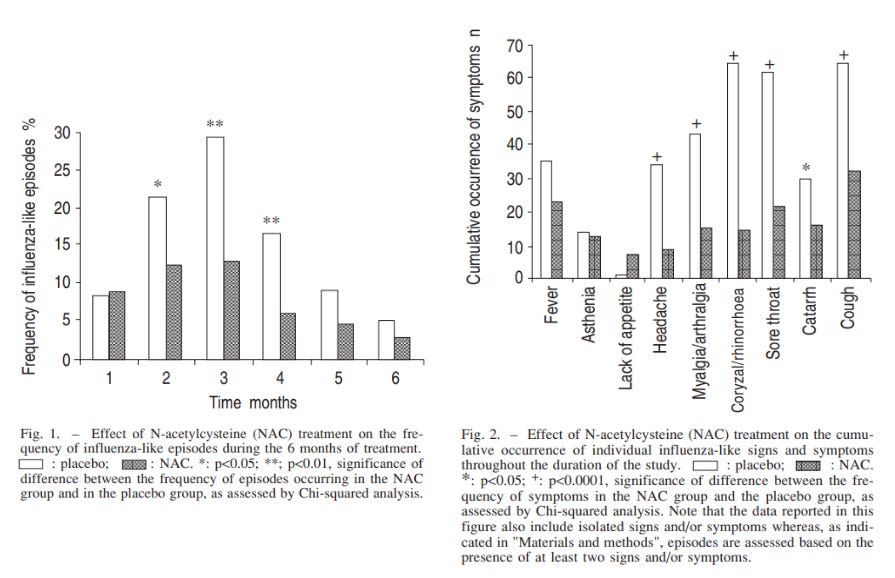
From De Flora, et. al. Fig. 1 (left) and Fig 2 (right) show rates of influenza-like symptoms and number of symptoms, respectively. In both instances, the NAC treated group (black bars) showed lower rates of symptoms and fewer symptoms in general compared to the placebo group (white bars). Note, however, that these results were contingent upon a high degree of self-reporting, and therefore may suffer from lack of transparency on the participant’s parts.
Interestingly, the researchers measured cell-mediated immunity2 as well using a test similar to an allergy test, such that participants had their skin exposed to 7 different antigens to see if there would be a reaction (i.e. rash or other skin alterations). Given these limitations, the researchers found that long-term use of NAC increased the number of immunological competent (normoergic- their skin reacted to the antigen) participants and attenuated the number of nonresponsive (anergic-their skin did not respond) participants.
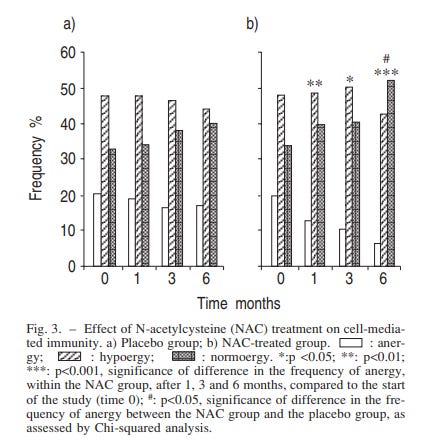
The results of this study are interesting, as both the NAC group and the placebo group started with the same cell-mediated response at month 0, suggesting that NAC may have helped improve cell-mediated immunity by the end of the 6 month assessment.
Overall, the results of this study provided some of the first looks into NAC as a possible prophylactic against respiratory infections. You’ve likely seen this study mentioned in reference to the 25% of NAC participants who experienced influenza-like symptoms compared to the 79% in the placebo group3. I've seen it referenced in many online articles as an argument in favor of NAC use for respiratory infections because of these findings.
As a caveat, note that some of the reporting provided to the researchers are heavily subjective and dependent upon the participant’s self-reporting:
Each subject was supplied with a personal diary card, and instructed to fill in any sign and/or symptom suggestive of an influenza-like episode, based on a suitable check list. Influenza-like episodes were assessed based on the presence of two or more of the following signs and/or symptoms: fever (≥38°C), asthenia, lack of appetite, headache, myalgia/arthralgia, coryza/rhynorrhoea, sore throat, catarrh and cough. At monthly intervals, the clinicians evaluated the diary card and performed a complete medical examination. The patients were also asked to contact the clinicians soon after the onset of any sign or symptom, and, whenever necessary, additional clinical examinations were performed.
As such, there’s a possibility that some patients may not have reported their symptoms which may have altered their symptomatic status (i.e. wrongly categorized them as asymptomatic, seroconverted). This is a general consequence of studies that rely on self-reporting since it’s hard to discern how transparent participants are being in their note-taking or awareness of their own symptoms.
However, given these results there was some founded hopes that NAC may serve as a useful prophylactic for influenza, and that use may be broadened to other respiratory illnesses such as COVID.
N-acetylcysteine improves oxidative stress and inflammatory response in patients with community acquired pneumonia (Zhang, et. al.)
A more recent study from Zhang, et. al.4 looked at the role of NAC in attenuating oxidative damage and the inflammatory response from pneumonia-like illness.
Oxidative damage and inflammation are intrinsically tied, and it’s assumed that these two are likely to lead to pulmonary damage during a case of pneumonia. Therefore, attenuating both should, in theory, lead to improved outcomes with reduced organ damage.
Patients who were diagnosed with pneumonia were randomized to either a group that was provided standard of care5 (control group) or one that was provided standard of care along with 600 mg of NAC twice a day (1200mg/day) for 10 days.
The diagnostic and inclusionary criteria were as follows:
The CAP diagnostic criteria were as follows: an acute pulmonary infiltrate evident on chest radiography and consistent with pneumonia; symptoms of a lower respiratory tract infection: fever, cough, and purulent sputum, with confirmatory findings of a clinical examination; acquisition of the infection outside a hospital, long-term care facility, or nursing home. The patients were included if they had the diagnostic criteria defined above, were diagnosed with bacterial community-acquired pneumonia according to American Thoracic Society/Infectious Diseases Society of America (ATS/IDSA) Guidelines[13,14] and if they were aged ≥18 years.
Because this study wanted to look at NAC in attenuating oxidative damage and inflammation, the researchers measured biomarkers from participants to see if there would be indications of improvement6:
The primary endpoint indicators were the changes in oxidative stress parameters (MDA, TAOC, SOD) and TNF-a after treatment in the NAC group compared with those in the nonNAC group. The secondary endpoint indicator was whether there was a difference in CT scores after treatment in the NAC group compared with the non-NAC group.
Relevant biomarkers were measured at the start of the study as well as at the 7 day mark. Improvements to biomarkers between the NAC group and the standard-of-care group showed that patients provided NAC showed greater improvement measures of the oxidative biomarker MDA as well as the inflammatory cytokine TNF-α relative to the non-NAC group. Measures of TAOC also appeared to have improved in the NAC group to a higher degree, however for SOD measures there didn’t appear to be significant improvements compared to the non-NAC group:
After 7 days plasma levels of MDA and TNF-a decreased more (MDA: p 0.004, TNF-a <0.001) in the NAC group than the non-NAC group, and there was a reliable increase of TAOC content in the NAC group (p 0.005). An improvement was seen in plasma SOD activity after treatment, but there was no significant difference in the rise of plasma SOD activity between groups (p 0.368). The results are shown in Table 3 and Fig. 2. No side effects were reported with NAC administration.
<0.001) in the NAC group than the non-NAC group, and there was a reliable increase of TAOC content in the NAC group (p 0.005). An improvement was seen in plasma SOD activity after treatment, but there was no significant difference in the rise of plasma SOD activity between groups (p 0.368). The results are shown in Table 3 and Fig. 2. No side effects were reported with NAC administration.
 <0.001) in the NAC group than the non-NAC group, and there was a reliable increase of TAOC content in the NAC group (p 0.005). An improvement was seen in plasma SOD activity after treatment, but there was no significant difference in the rise of plasma SOD activity between groups (p 0.368). The results are shown in Table 3 and Fig. 2. No side effects were reported with NAC administration.
<0.001) in the NAC group than the non-NAC group, and there was a reliable increase of TAOC content in the NAC group (p 0.005). An improvement was seen in plasma SOD activity after treatment, but there was no significant difference in the rise of plasma SOD activity between groups (p 0.368). The results are shown in Table 3 and Fig. 2. No side effects were reported with NAC administration.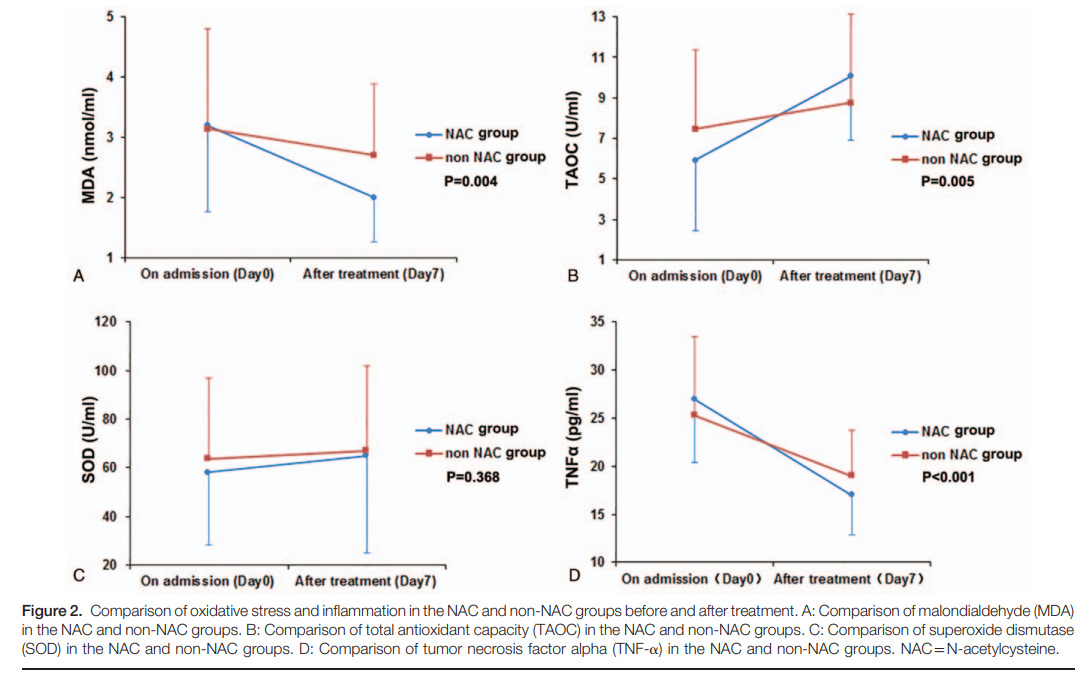
Figure 2. The above graphs map out biomarkers relevant to oxidative and inflammatory responses. Note that the oxidative molecule MDA and the inflammatory cytokine TNF-α should decrease, and here it appears that the NAC group (in blue) showed a higher decline in MDA and TNF-α levels compared to the non NAC (control) group. For TAOC there should be an expectation that
Interestingly, CT scan results, which were used to measure improvements with pneumonia, showed no statistically significant improvements in the NAC group compared to the non-NAC group7, inferring that pulmonary damage and pneumonia may not be alleviated through NAC use.
So although this study did not show greater improvement scores for NAC patients, the results do suggest that NAC may reduce oxidative stress and inflammation caused by pneumonia.
What’s important from this study is that the researchers used a highly accessible form of NAC (tablets) which would be easy to administer at an at-home environment, similar to the one given in the De Flora, et. al. study8.
However, it’s worth noting that this study used a very small sample size, likely driven by issues in recruitment9:
Initially 86 patients were considered for inclusion in the study. Twenty-nine patients were excluded and so 37 cases were in the NAC group and 24 cases in the non-NAC group. Eighteen cases did not complete the study. So, 39 pneumonia patients completed the study. Of these, 21 subjects were included in the NAC group and 18 in the non-NAC group.
This may have altered results due to not having the eligibility necessary for meeting effect size.
Also, note that the researchers never examined the etiological agent responsible for pneumonia, such that there was no information as to whether the pneumonia was caused by a viral, bacterial, or fungal infection. The researchers do rely on CT scans for diagnosis, as well as if participants met the guidelines for bacterial pneumonia from the American Thoracic Society/Infectious Diseases Society of America (ATS/IDSA):
Another limitation is that the study did not record details of the etiologic agent. Patients in our study were diagnosed with bacterial community-acquired pneumonia according to ATS/IDSA Guidelines. For the pathological features, treatment and disease course of tuberculosis and fungus infection are different from those of bacterial pneumonia, patients considered to have tuberculosis or fungus infection were not included. Primary viral pneumonia was defined in patients presenting during the acute phase of influenza virus illness with acute respiratory disease and unequivocal alveolar opacification involving ≥2 lobes with negative respiratory and blood bacterial cultures. There might be some differences in oxidative stress status between bacterial and viral pneumonia.[26] Primary viral pneumonia cases were not admitted to the study either.
Since different infections are likely to lead to different levels of oxidative damage and inflammation, we should keep in mind that the results here are likely to be influenced by the type of pathogen i.e. no direct relationship to viral pneumonia in this study.
In short, these two studies- along with a few others not mentioned here- provide much of the basis for examining NAC within the context of COVID. Given these results it’s no wonder why NAC has been looked at, and considering that NAC is widely available (or at least used to be) it makes sense why research continues to look into NAC as a possible prophylactic and therapeutic against COVID.
[continued next post]
Last edited: